4. Molecular mechanisms of salt stress response
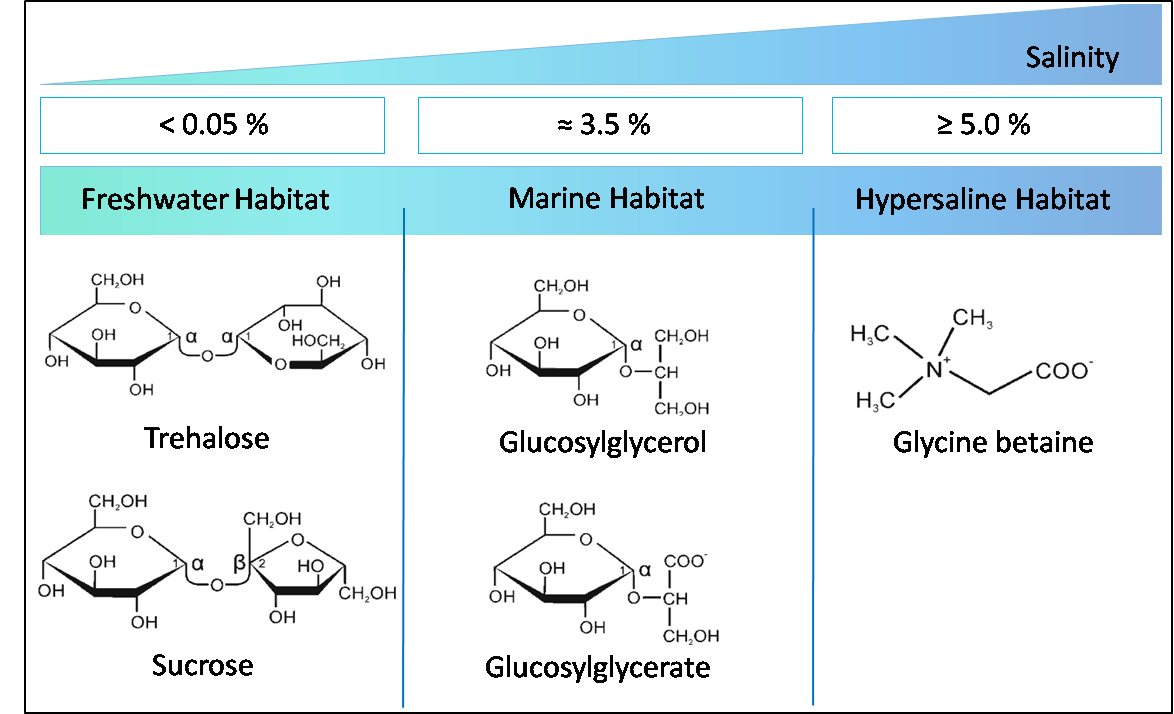
Cyanobacteria can be found in all photic habitats of Earth. They thrive in waters of different salt contents, from freshwater over marine systems to hypersaline ponds. To acclimate to enhanced salinities, cyanobacterial cell export toxic inorganic ions and accumulate compatible solutes. The kind of preferred compatible solute obviously determines the final salt tolerance, since a close correlation between compatible solute and salt tolerance has been observed (Hagemann 2011). However, deviations from this rule have been reported (Pade et al. 2016). The ability of various cyanobacteria to tolerate high and/or fluctuating salinities attenuates the requirement of freshwater in their mass cultivation, which makes these organisms even more interesting for a sustainable production of chemical compounds from sunlight and CO2 (Kirsch et al. 2019).
The model organism Synechocystis sp. PCC 6803 and many marine strains respond to increasing salinities mainly by the accumulation of glucosylglycerol (GG) and sucrose. The accumulation of the compatible solute GG is achieved by de novo synthesis. The key target of regulation is the enzyme GG-phosphate synthase (GgpS) and involves transcriptional, posttranscriptional and biochemical mechanisms. The GG-degrading enzyme GG hydrolase A (GghA) is particularly important for GG degradation during exposure to decreasing salinities. The inversely ion-regulated activities of GgpS and GghA could represent the main model for tuning the GG steady-state levels according to external salinities (Kirsch et al. 2019). In addition to its role as stress protectant, GG and other compatible solutes are also promising targets for biotechnology.
In our group we are currently interested how cyanobacteria sense external salinities. Until now some tw-component regulatory systems have been identified, which are involved in the sensing of general stress among cyanobacteria (Marin et al. 2003), but a specific cyanobacterial salt sensor still awaits identification in the future. Moreover, cyanobacterial genes/proteins can be used to improve the salt and drought tolerance of model plants such as Arabidopsis (Klähn et al 2009) as well as crop plants such as potato (Sievers et al. 2013).
References
Hagemann M (2011) Molecular biology of cyanobacterial salt acclimation. FEMS Microbiology Reviews 35:87-123
Kirsch F, Klähn S, Hagemann M (2019) Salt-regulated accumulation of the compatible solutes sucrose and glucosylglycerol in cyanobacteria and its biotechnological relevance. Frontiers in Microbiology 10:2139
Klähn S, Marquardt DM, Rollwitz I, Hagemann M (2009) Expression of the ggpPS gene for glucosylglycerol biosynthesis from Azotobacter vinelandii improves the salt tolerance of Arabidopsis thaliana. Journal of Experimental Botany 60:1679-1689
Marin K, Suzuki I, Yamaguchi K, Ribbeck K, Yamamoto H, Kanesaki Y, Hagemann M, Murata N (2003) Identification of histidine kinases that act as sensors in the perception of salt stress in Synechocystis sp. strain PCC 6803. Proceeding of the National Academy of Sciences USA 100:9061-9066
Pade N, Michalik D, Ruth W, Belkin N, Hess WR, Berman-Frank I, Hagemann M (2016) Trimethylated homoserine functions as the major compatible solute in the globally significant oceanic cyanobacterium Trichodesmium. Proceeding of the National Academy of Sciences USA 113:13191-13196
Sievers N, Muders K, Henneberg M, Klähn S, Effmert M, Junghans H, Hagemann M (2013) Establishing glucosylglycerol synthesis in potato (Solanum tuberosum L. cv. Albatros) by expression of the ggpPS gene from Azotobacter vinelandii. Journal of Plant Science and Molecular Breeding, doi:10.7143/2050-2389-2-1